  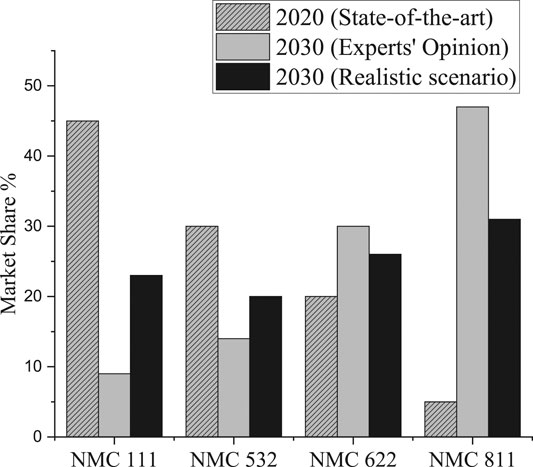
1–3 However, batteries using these Ni-rich NMCs and a graphite negative electrode suffer from rapid capacity fade, limiting the lifetime of the battery. 
Characterisation of NMC/graphite “full cells” reveals that many of the products formed at the NMC electrode are consumed at the negative electrode, emphasising the importance of studying electrode cross-talk processes to understand the origin of capacity loss in NMC/graphite batteries.ġ Introduction The use of Ni-rich layered transition metal oxides ( e.g., LiNi xMn 圜o 1− x− yO 2, NMC) as positive electrode materials in lithium-ion battery packs is favoured over LiCoO 2 due to their higher energy densities, and because cobalt, with its toxicity, cost and mining issues, is largely replaced with nickel. Two main and distinct decomposition mechanisms are identified, each with a different onset voltage, and very different decomposition products. We generate 17O-labelled singlet oxygen ( 1O 2) photochemically and use it to investigate proposed electrolyte decomposition pathways. In the present work, we characterise the electrolyte decomposition products formed at LiNi xMn 圜o 1− x− yO 2 (NMC) positive electrodes using solution NMR spectroscopy and operando gas measurements. One major cause of the loss in capacity of these batteries is linked with the decomposition of the electrolyte solution, and the subsequent reactions of these products at the negative electrode, a process referred to as “electrode cross-talk”. Ni-rich layered positive electrode materials are now universally used in electric vehicle batteries and yet their degradation pathways are still not understood. Broader context The development of rechargeable batteries with longer lifetimes represents a major challenge in enabling the shift from fossil fuel-powered to electric vehicles. The insights on electrolyte decomposition mechanisms at the positive electrode, and the consumption of decomposition products at the negative electrode contribute to understanding the origin of capacity loss in NMC/graphite cells, and are hoped to support the development of strategies to mitigate the degradation of NMC-based cells. Noticeably fewer decomposition products were detected in NMC/graphite cells compared to NMC/Li xFePO 4 cells, which is ascribed to the consumption of water (from the reaction of 1O 2 and EC) at the graphite electrode, preventing secondary decomposition reactions. The water that is produced initiates secondary reactions, leading to the formation of the various products identified by NMR spectroscopy. The formation of water via this mechanism was confirmed by reacting 17O-labelled 1O 2 with EC and characterising the reaction products via 1H and 17O NMR spectroscopy. At low potentials (80% SOC), 1O 2 released from the transition metal oxide chemically oxidises the electrolyte solvent (EC) to form CO 2, CO and H 2O. Based on the detected soluble and gaseous products, two distinct routes with different onset potentials are proposed for the decomposition of the electrolyte solution at NMC electrodes. A partially delithiated LFP (Li xFePO 4) counter electrode was used to selectively identify the products formed through processes at the positive electrodes. In this work, a combination of operando gas measurements and solution NMR was used to study decomposition reactions of the electrolyte solution at NMC (LiNi xMn 圜o 1− x− yO 2) and LCO (LiCoO 2) electrodes. However, the exact mechanism(s) for electrolyte decomposition at the positive electrode, and particularly the soluble decomposition products that form and initiate further reactions at the negative electrode, are still largely unknown. 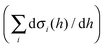
Preventing the decomposition reactions of electrolyte solutions is essential for extending the lifetime of lithium-ion batteries. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
